Europium - 63Eu: properties of free atoms
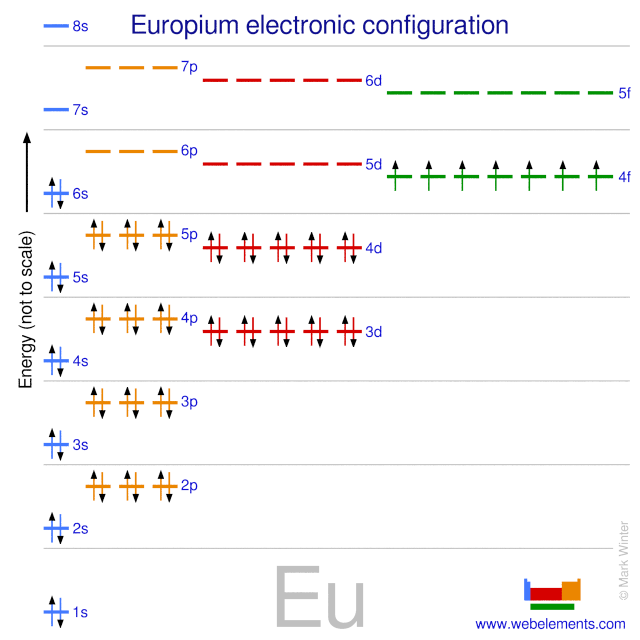
Europium atoms have 63 electrons and the shell structure is 2.8.18.25.8.2.
The ground state electron configuration of ground state gaseous neutral europium is [Xe].4f7.6s2 and the term symbol is 8S7/2.

Atomic spectrum
A representation of the atomic spectrum of europium.
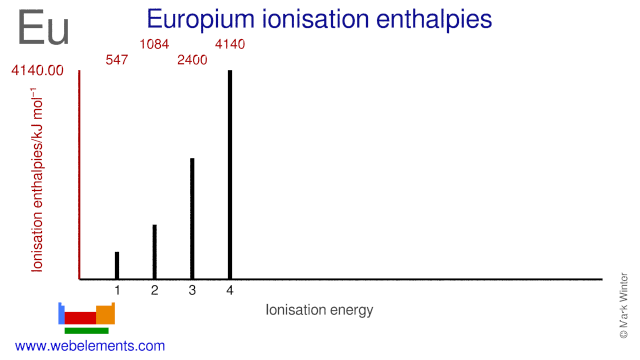
Ionisation Energies and electron affinity
The electron affinity of europium is < 50 0.5 eV kJ mol‑1. The ionisation energies of europium are given below.
Effective Nuclear Charges
The following are "Clementi-Raimondi" effective nuclear charges, Zeff. Follow the hyperlinks for more details and for graphs in various formats.
| 1s | 61.76 | ||||||
|---|---|---|---|---|---|---|---|
| 2s | 46.47 | 2p | 58.72 | ||||
| 3s | 42.87 | 3p | 43.28 | 3d | 49.25 | ||
| 4s | 32.87 | 4p | 31.87 | 4d | 28.94 | 4f | 24.32 |
| 5s | 18.59 | 5p | 16.56 | 5d | (no data) | ||
| 6s | 8.12 | 6p | (no data) | ||||
| 7s | |||||||
References
These effective nuclear charges, Zeff, are adapted from the following references:
- E. Clementi and D.L.Raimondi, J. Chem. Phys. 1963, 38, 2686.
- E. Clementi, D.L.Raimondi, and W.P. Reinhardt, J. Chem. Phys. 1967, 47, 1300.
Electron binding energies
| Label | Orbital | eV [literature reference] |
|---|---|---|
| K | 1s | 48519 [1] |
| L I | 2s | 8052 [1] |
| L II | 2p1/2 | 7617 [1] |
| L III | 2p3/2 | 6977 [1] |
| M I | 3s | 1800 [1] |
| M II | 3p1/2 | 1614 [1] |
| M III | 3p3/2 | 1481 [1] |
| M IV | 3d3/2 | 1158.6 [2] |
| M V | 3d5/2 | 1127.5 [2] |
| N I | 4s | 360 [1] |
| N II | 4p1/2 | 284 [1] |
| N III | 4p3/2 | 257 [1] |
| N IV | 4d3/2 | 133 [1] |
| N V | 4d5/2 | 127.7 [2] |
| N VI | 4f5/2 | 0 [1] |
| N VII | 4f7/2 | 0 [1] |
| O I | 5s | 32 [1] |
| O II | 5p1/2 | 22 [1] |
| O III | 5p3/2 | 22 [1] |
Notes
I am grateful to Gwyn Williams (Jefferson Laboratory, Virginia, USA) who provided the electron binding energy data. The data are adapted from references 1-3. They are tabulated elsewhere on the WWW (reference 4) and in paper form (reference 5).
References
- J. A. Bearden and A. F. Burr, "Reevaluation of X-Ray Atomic Energy Levels," Rev. Mod. Phys., 1967, 39, 125.
- M. Cardona and L. Ley, Eds., Photoemission in Solids I: General Principles (Springer-Verlag, Berlin) with additional corrections, 1978.
- Gwyn Williams WWW table of values
- D.R. Lide, (Ed.) in Chemical Rubber Company handbook of chemistry and physics, CRC Press, Boca Raton, Florida, USA, 81st edition, 2000.
- J. C. Fuggle and N. Mårtensson, "Core-Level Binding Energies in Metals," J. Electron Spectrosc. Relat. Phenom., 1980, 21, 275.