| The University of Sheffield |
| Department of Chemistry |
VSEPR |
VSEPR calculation for perchlorate, [ClO4]-
Perchlorate, [ClO4]-
| Lewis structure: |
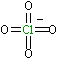 |
| Central atom: |
chlorine |
| Valence electrons on central atom: |
7 |
| 4 terminal oxygens each contribute 1 electron in the four σ bonds |
4 |
| Subtract four for the four electrons contributed by Cl to the four π bonds (one for each): |
-4 |
| Add one for the negative charge located on Cl |
1 |
| Total: |
8 |
| Divide by 4 to give electron pairs |
4 |
| 4 electron pairs: |
tetrahedral geometry for the four shape-determining electron pairs |
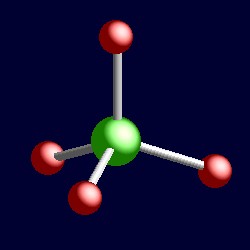 |
|
| The geometry of perchlorate, [ClO4]-. You can use your mouse to manipulate the molecule in the right hand "Jmol" image. |
|



