 |
|---|
| VSEPR home |
|
|
| The University of Sheffield | |
| Department of Chemistry | VSEPR |
Geometries
It is necessary to know the most favourable arrangement for any given number of electron pairs surrounding any particular atom. These arrangements are found using simple geometrical constructions. This involves placing the nucleus of the atom in question at the centre of a sphere and then placing the electron pairs on the surface of the sphere so that they are as far apart as possible. The resulting arrangements are often intuitively obvious.
| Electron pairs | Geometry |
|---|---|
| 2 | linear |
| 3 | trigonal planar |
| 4 | tetrahedral |
| 5 | trigonal bipyramidal |
| 6 | octahedral |
For the case of just two electron pairs, the arrangement is simple and the minimum energy configuration is when the electron pairs form a linear arrangement with the nucleus. In this configuration the electron pair-nucleus-electron pair angle is 180°. The coordination geometry of the central atom is described as linear.
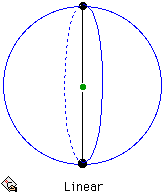
Arrangement for 2 electron pairs on the surface of a sphere.
Three electron pairs arrange themselves trigonally, that is with bond angles of 120°. For four electron pairs, one might expect the square-planar geometry to be favourable. However, tetrahedral bond angles are 109.5°, larger than the square-planar angles of 90°. If, for the purposes of illustration, the electron pairs are assumed to be points then it should be clear that, for similar bond lengths, the electron pairs are necessarily further apart in the tetrahedral arrangement than in a square planar arrangement. There is more electron pair-electron pair repulsion in the square-planar geometry and so the tetrahedral geometry is favoured.
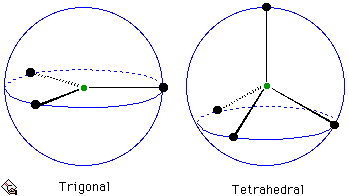
Arrangements for 3 and 4 electron pairs on the surface of a sphere.
The case of five coordination is a little trickier. Most molecules whose shape is determined by five electron pairs are trigonal bipyramidal.
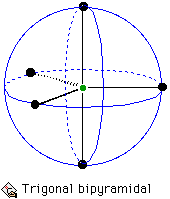
Arrangement for 5 electron pairs on the surface of a sphere.
There are two environments in a trigonal bipyramid, axial and equatorial. These two environments are chemically distinct. There is another very reasonable candidate, and that is the square-based pyramid. In effect this arrangement is an octahedron in which one group is removed and in which the four adjacent groups move down slightly to occupy partially the resulting vacancy. In practice, this geometry is only a little disfavoured relative to the trigonal bipyramid and the square-based pyramidal geometry is very important in the interconversion of axial and equatorial environments in trigonal bipyramids.
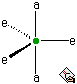
Axial and equatorial environments for the trigonal bipyramid.
For six-coordinate systems, the octahedral geometry is by far the most important. An alternative geometry, the trigonal prism, is uncommon.
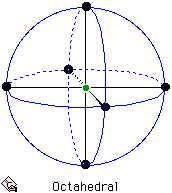
Arrangement for 6 electron pairs on the surface of a sphere.
 |
|---|
|
|