| The University of Sheffield |
| Department of Chemistry |
VSEPR |
VSEPR calculation for chlorine trifluoride, ClF3
Refer to the previous section for a discussion on the isomer possibilities for this compound.
Chlorine trifluoride, ClF3
| Lewis structure: |
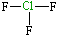 |
| Central atom: |
chlorine |
| Valence electrons on central atom: |
7 |
| 3 F atoms each contribute 1 electron: |
3 |
| Total: |
10 |
| Divide by 2 to give electron pairs |
5 |
| 5 electron pairs: |
trigonal bipyramidal geometry for the five shape-determining electron pairs |
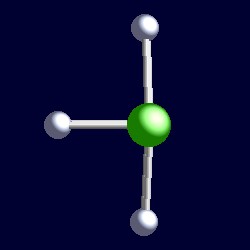 |
|
| The geometry of chlorine trifluoride, ClF3. You can use your mouse to manipulate the molecule in the right hand "Jmol" image. |
| 

