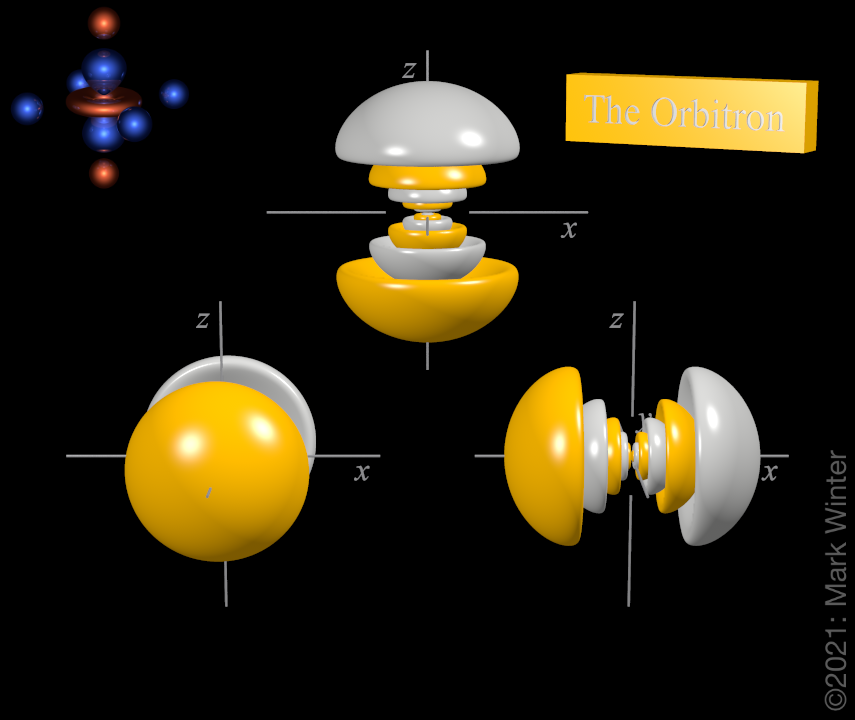
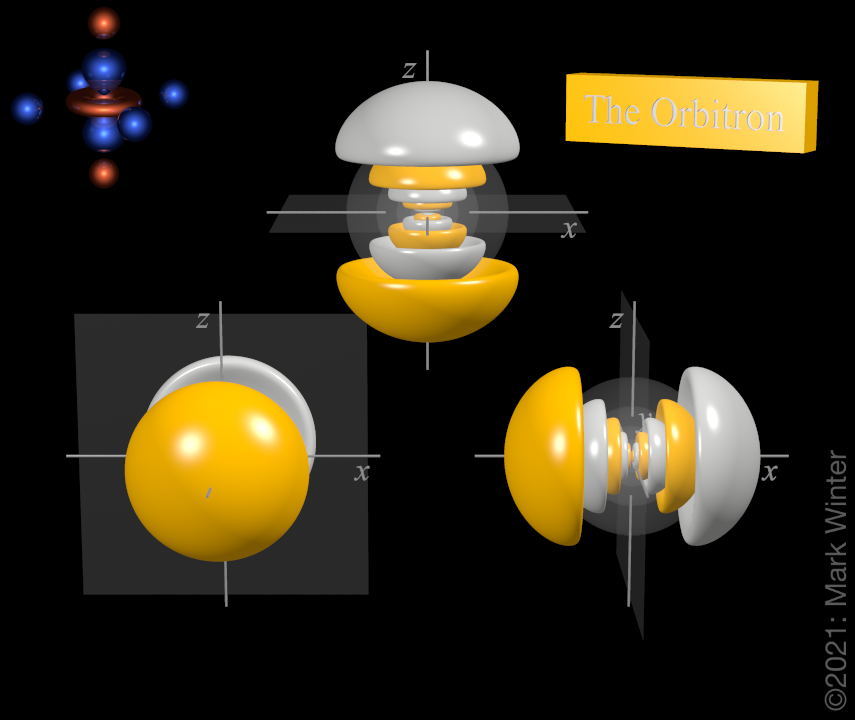
7p orbitals
The shape of the three 7p orbitals. From left to right: 7py, 7pz, and 7px. For each, the yellow zones are where the wave functions have positive values and the white zones denote negative values.
For any atom, there are three 7p orbitals. These orbitals have the same shape but are aligned differently in space. The three 7p orbitals normally used are labelled 7px, 7py, and 7pz since the functions are "aligned" along the x, y, and z axes respectively.
Each 7p orbital has 12 lobes. There is a planar node normal to the axis of the orbital (so the 7px orbital has a yz nodal plane, for instance). Apart from the planar node there are also five spherical nodes that partition off the small inner lobes.
The origin of the planar node becomes clear when inspecting the wave equations which, for instance, includes an x term in the case of the 2px orbital. When x = 0, then there is a node, and this by definition is the yz plane.
The origin of the spherical nodes becomes clearer if we examine the wave equations, which include a (6720 - 8400ρ + 3360ρ2 - 420ρ3 + 40ρ4 - ρ5) term. When (6720 - 8400ρ + 3360ρ2 - 420ρ3 + 40ρ4 - ρ5) = 0, then there are nodes.
The OrbitronTM, a gallery of orbitals on the WWW: https://winter.group.shef.ac.uk/orbitron/
Copyright 2002-2023 Prof. Mark Winter [The University of Sheffield]. All rights reserved.